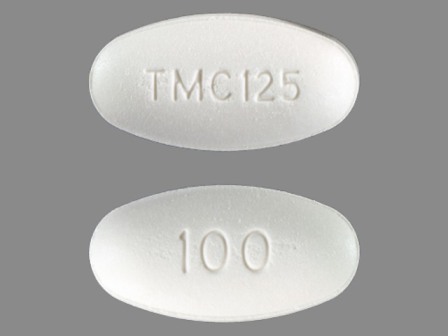
Intelence
Active Ingredient(s): EtravirineFDA Approved: * January 18, 2008
Pharm Company: * TIBOTEC
Category: HIV / AIDS
Etravirine (ETR,[1] brand name Intelence, formerly known as TMC125) is a drug used for the treatment of HIV. Etravirine is a non-nucleoside reverse transcriptase inhibitor (NNRTI). Unlike the currently available agents in the class, resistance to other NNRTIs does not seem to confer resistance to etravirine.[2] Etravirine is marketed by Janssen, a subsidiary of Johnson & Johnson. In January 2008, the Food and Drug Administration approved its use for pati... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.Dosage List