Ranitidine - NDC Database (Page 3)
564 records found
Start a Discussion
42708-118 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by Qpharma Inc
43063-087 Dec 16, 2009
Ranitidine 300 mg (Ranitidine Hydrochloride 336 mg) Oral Tablet by Pd-rx Pharmaceuticals, Inc.
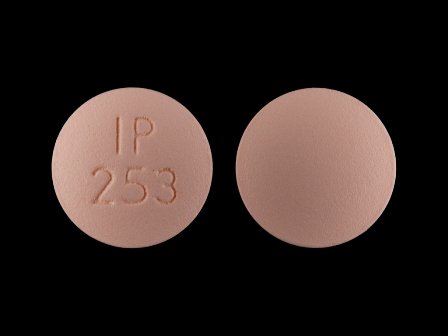
43063-844 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by Pd-rx Pharmaceuticals, Inc.
43353-967 Aug 18, 2014
Ranitidine 300 mg Oral Tablet by Aphena Pharma Solutions - Tennessee, LLC
43353-971 Aug 18, 2014
Ranitidine 150 mg Oral Tablet by Aphena Pharma Solutions - Tennessee, LLC
45865-416 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by Medsource Pharmaceuticals
49349-476 Aug 11, 2011
Ranitidine 150 mg (As Ranitidine Hydrochloride 168 mg) Oral Tablet by Remedyrepack Inc.
49999-043 Oct 06, 2010
Ranitidine 150 mg Oral Tablet by Lake Erie Medical Dba Quality Care Products LLC
49999-428 Nov 19, 2008
Ranitidine 300 mg Oral Tablet, Film Coated by Lake Erie Medical Dba Quality Care Products LLC
50090-0604 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by A-s Medication Solutions
50090-0605 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by A-s Medication Solutions
50090-1496 Nov 19, 2008
Ranitidine 300 mg Oral Tablet, Film Coated by A-s Medication Solutions
50090-1896 Aug 29, 1997
Ranitidine Hydrochloride 150 mg Oral Tablet, Film Coated by A-s Medication Solutions
50090-2574 Nov 19, 2008
Ranitidine 150 mg Oral Tablet, Film Coated by A-s Medication Solutions
50090-2740 Nov 19, 2008
Ranitidine 150 mg Oral Tablet, Film Coated by A-s Medication Solutions
50268-692 Aug 04, 2015
Ranitidine 150 mg Oral Tablet by Avpak
50436-0253 Dec 16, 2009
Ranitidine 150 mg Oral Tablet by Unit Dose Services
50436-6930 Nov 19, 2008
Ranitidine 150 mg (As Ranitidine Hydrochloride 168 mg) Oral Tablet by Unit Dose Services
50436-6942 Nov 19, 2008
Ranitidine 300 mg (Ranitidine Hydrochloride 336 mg) Oral Tablet by Unit Dose Services
51655-681 Sep 15, 2014
Ranitidine 150 mg Oral Tablet by Northwind Pharmaceuticals, LLC
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us .