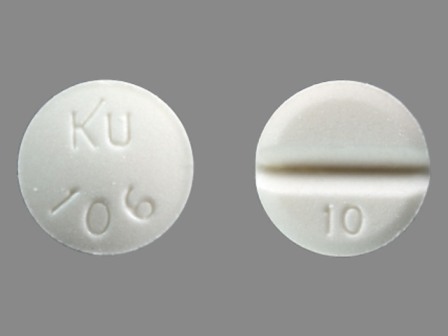
Isosorbide Mononitrate - NDC Database (Page 5)

Isosorbide Mononitrate 120 mg Oral Tablet, Extended Release by Remedyrepack Inc.

Isosorbide Mononitrate 60 mg Oral Tablet, Extended Release by Remedyrepack Inc.

Isosorbide Mononitrate 60 mg Oral Tablet, Extended Release by Denton Pharma, Inc. Dba Northwind Pharmaceuticals
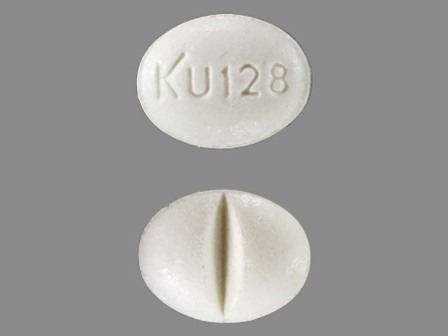
Isosorbide Mononitrate 30 mg Oral Tablet, Extended Release by Denton Pharma, Inc. Dba Northwind Pharmaceuticals

Isosorbide Mononitrate 60 mg Oral Tablet, Extended Release by Bryant Ranch Prepack
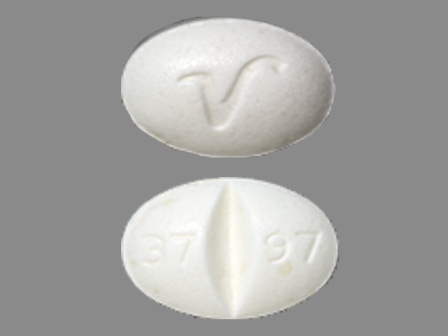
Isosorbide Mononitrate 30 mg Oral Tablet, Extended Release by Bryant Ranch Prepack

Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Mckesson Contract Packaging
0143-2230 Mar 30, 2006
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by West-ward Pharmaceutical Corp
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by West-ward Pharmaceutical Corp
0228-2711 Mar 31, 2007
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Actavis Elizabeth LLC
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Actavis Elizabeth LLC
0228-2713 Mar 31, 2007
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by Actavis Elizabeth LLC
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by Actavis Elizabeth LLC
0603-4111 Aug 11, 2010
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Qualitest Pharmaceuticals
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Qualitest Pharmaceuticals
0615-4546 Mar 30, 2006
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 30 mg 24 Hr Extended Release Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
0615-5549 Oct 30, 1998
Isosorbide Mononitrate 10 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 10 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
0615-5550 Oct 30, 1998
Isosorbide Mononitrate 20 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 20 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
0615-7757 Aug 11, 2010
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 60 mg 24 Hr Extended Release Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
0615-8043 Dec 19, 2013
Isosorbide Mononitrate 30 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 30 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
0615-8044 Dec 19, 2013
Isosorbide Mononitrate 60 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Isosorbide Mononitrate 60 mg Oral Tablet by Ncs Healthcare of Ky, Inc Dba Vangard Labs
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.