Benazepril Hydrochloride - NDC Database
241 records found
Page: 1 Start a Discussion
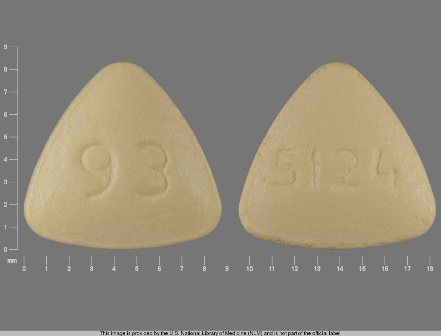
0093-5124 Feb 11, 2004
Bzp Hydrochloride 5 mg Oral Tablet by Teva Pharmaceuticals USA Inc
0093-5125 Feb 11, 2004
Bzp Hydrochloride 10 mg Oral Tablet by Teva Pharmaceuticals USA Inc
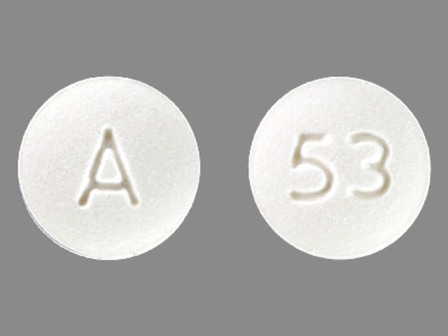
0093-5126 Feb 11, 2004
Bzp Hydrochloride 20 mg Oral Tablet by Teva Pharmaceuticals USA Inc
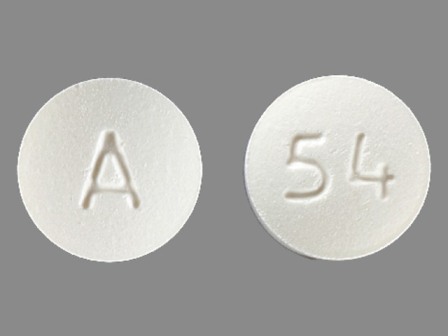
0093-5127 Feb 11, 2004
Bzp Hydrochloride 40 mg Oral Tablet by Teva Pharmaceuticals USA Inc
0185-0048 Feb 11, 2004
Bzp Hydrochloride 40 mg Oral Tablet by Eon Labs, Inc.
0185-0053 Feb 11, 2004
Bzp Hydrochloride 10 mg Oral Tablet by Eon Labs, Inc.
0185-0505 Feb 11, 2004
Bzp Hydrochloride 5 mg Oral Tablet by Eon Labs, Inc.
0185-0820 Feb 11, 2004
Bzp Hydrochloride 20 mg Oral Tablet by Eon Labs, Inc.
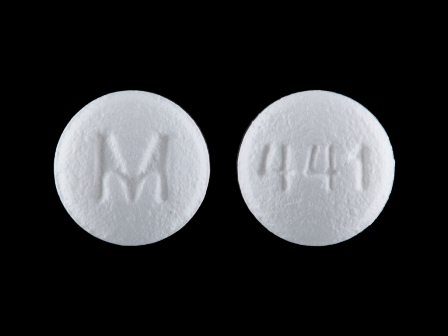
0378-0441 Dec 03, 2012
Bzp Hydrochloride 5 mg Oral Tablet by Mylan Pharmaceuticals Inc.
13811-627 Nov 15, 2012
Bzp Hydrochloride 5 mg Oral Tablet by Trigen Laboratories, Inc.
42291-160 Jul 29, 2009
Bzp Hydrochloride 5 mg Oral Tablet by Avkare, Inc.
42291-161 Jul 29, 2010
Bzp Hydrochloride 10 mg Oral Tablet by Avkare, Inc.
42291-162 Jul 29, 2009
Bzp Hydrochloride 20 mg Oral Tablet by Avkare, Inc.
42291-163 Jul 29, 2009
Bzp Hydrochloride 40 mg Oral Tablet by Avkare, Inc.
43063-131 Sep 29, 2011
Bzp Hydrochloride 20 mg Oral Tablet by Pd-rx Pharmaceuticals, Inc.
43063-132 Feb 11, 2004
Benazepril Hydrochloride 40 mg/1 Oral Tablet, Coated by Pd-rx Pharmaceuticals, Inc.
43547-335 Jan 01, 2014
Benazepril Hydrochloride 5 mg/1 Oral Tablet, Coated by Solco Healthcare U.S., LLC
43547-336 Jan 01, 2014
Benazepril Hydrochloride 10 mg/1 Oral Tablet, Coated by Solco Healthcare U.S., LLC
43547-337 Jan 01, 2014
Benazepril Hydrochloride 20 mg/1 Oral Tablet, Coated by Solco Healthcare U.S., LLC
43547-338 Jan 01, 2014
Benazepril Hydrochloride 40 mg/1 Oral Tablet, Coated by Solco Healthcare U.S., LLC
Page: 1 Start a Discussion
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us .