Mavik
Active Ingredient(s): TrandolaprilFDA Approved: * April 26, 1996
Pharm Company: * ABBOTT
Category: Blood Pressure
Trandolapril is an ACE inhibitor used to treat high blood pressure. It may also be used to treat other conditions. It is similar in structure to another ACE Inhibitor, Ramipril but has a cyclohexane group. It also is a pro-drug and must get metabolized. It has an extended half-life and therefore has a higher potency. It was patented in 1981, and approved for medical use in 1993.[1] It is marketed by Abbott Laboratories under the brand name Mavik. Contents 1 Side effects 2 ... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.2 Discussions
Dosage List

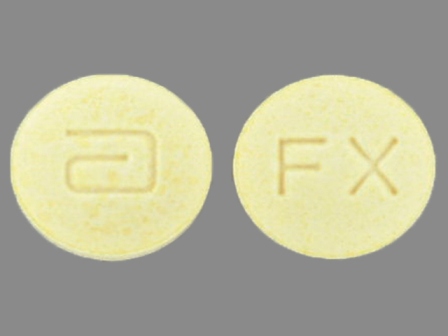
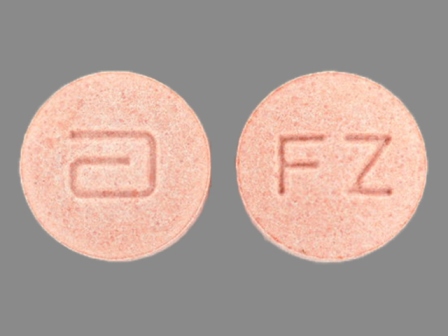
Popular Topics
mavik generic h03 side l u other side
round pink h03 side l u sidegeneric b/p...
mavik 2mg tablets
what is it for...