Edarbyclor
Active Ingredient(s): Azilsartan Medoxomil + ChlorthalidoneFDA Approved: * December 20, 2011
Pharm Company: * TAKEDA PHARMS NA
Category: Blood Pressure
Azilsartan is an angiotensin II receptor antagonist used in the treatment of hypertension,[1][2][3] developed by Takeda. It is marketed in tablet form under the brand name Edarbi as the prodrug azilsartan medoxomil.[4] The most common adverse reaction in adults is diarrhea.[1] It is also sold as a combination drug with chlortalidone under the brand name Edarbyclor.[5] Contents 1 Structure activit... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.6 Discussions
Dosage List
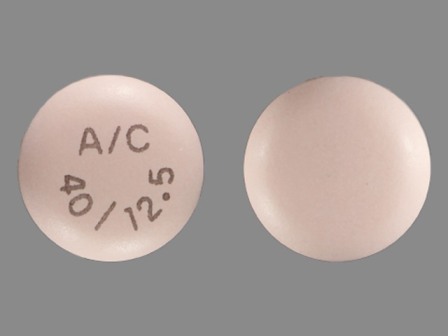



Popular Topics
I have had uncontrolled high bp 35 years. I am called hyper sodium sensitive and have been to many research drs. If you ...
How long does a Edarbyclor 40mg/12.5 mg tablet remain in your system? ## According to the FDA, the elimination half-live...
This med is amazing at bringing down by bp however I still feel like my head is foggy. My heart races to up 100. Does th...