69189-0242 : Toviaz 4 mg Oral Tablet, Film Coated, Extended Release
| NDC: | 69189-0242 |
| Labeler: | Avera Mckennan Hospital |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Toviaz Toviaz |
| Dosage Form: | Oral Tablet, Film Coated, Extended Release |
| Application #: | NDA022030 |
| Rev. Date: |
Appearance:
| Markings: | FS |
| Shapes: |
Oval |
| Colors: |
 Blue Blue |
| Size (mm): | 13 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
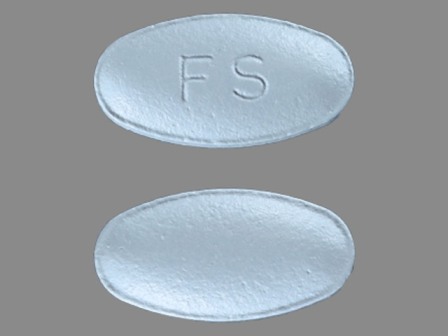
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 69189-0242-1: 1 TABLET, FILM COATED, EXTENDED RELEASE IN 1 DOSE PACK (69189‑0242‑1)
Active Ingredients:
- Fesoterodine Fumarate
Dosage Strength:
- 4 mg
Inactive Ingredients:
- Glyceryl Dibehenate
- Hypromelloses
- Indigotindisulfonate Sodium
- Aluminum Oxide
- Lactose Monohydrate
- Lecithin, Soybean
- Cellulose, Microcrystalline
- Polyethylene Glycols
- Polyvinyl Alcohol
- Talc
- Titanium Dioxide
- Xylitol
Related Products:
Based on records with the same trade name.- 69189-0244 Toviaz 8 mg Oral Tablet, Film Coated, Extended Release by Avera Mckennan Hospital
- 0069-0242 24 Hr Toviaz 4 mg Extended Release Tablet by Pfizer Laboratories Div Pfizer Inc
- 0069-0244 24 Hr Toviaz 8 mg Extended Release Tablet by Pfizer Laboratories Div Pfizer Inc
- 54868-6156 24 Hr Toviaz 4 mg Extended Release Tablet by Physicians Total Care, Inc.
- 54868-6175 24 Hr Toviaz 8 mg Extended Release Tablet by Physicians Total Care, Inc.
- 55154-2737 Toviaz 4 mg Oral Tablet, Film Coated, Extended Release by Cardinal Health
- 55154-2738 Toviaz 8 mg Oral Tablet, Film Coated, Extended Release by Cardinal Health
- 63539-183 24 Hr Toviaz 8 mg Extended Release Tablet by U.S. Pharmaceuticals
- 63539-242 24 Hr Toviaz 4 mg Extended Release Tablet by U.S. Pharmaceuticals
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 69189-0239Next: 69189-0243 >
Related Discussions:
Toviaz 4mg containing Fesoterodine Fumarate, Made by Pfizer Ltd.
Toviaz 4mg tabs were prescribed to me by my Urology Consultant after my TURP operation on 24th March 2014 while I was in...
Toviaz 4mg tabs were prescribed to me by my Urology Consultant after my TURP operation on 24th March 2014 while I was in...
Fesoterodine Fumarate vs Detrol LA and Diovan
Is Fesoterodine Fumarate essentially the same drug as Detrol LA and Diovan? ## Fesoteridine is the active ingredient in ... 1 reply
Is Fesoterodine Fumarate essentially the same drug as Detrol LA and Diovan? ## Fesoteridine is the active ingredient in ... 1 reply
Toviaz 4 mg every other day
Has anyone taken toviaz 4 mg every other day to reduce constipation and found it still provides relief? ## has anyone ta... 3 replies
Has anyone taken toviaz 4 mg every other day to reduce constipation and found it still provides relief? ## has anyone ta... 3 replies
toviaz quit working
After taking for 5 years, toviaz just quit working. Is it normal to build up a resistance? Will it start working again i... 2 replies
After taking for 5 years, toviaz just quit working. Is it normal to build up a resistance? Will it start working again i... 2 replies
Toviaz stops working
I have oab for about 3-4 years now and I have been take toviaz for that time. During my vacation I need to urinate a lot... 2 replies
I have oab for about 3-4 years now and I have been take toviaz for that time. During my vacation I need to urinate a lot... 2 replies
Toviaz stopped working after 4 weeks
I started taking Toviaz to help with my frequent urination. It worked great for about 4 weeks but then it lost its effec... 2 replies
I started taking Toviaz to help with my frequent urination. It worked great for about 4 weeks but then it lost its effec... 2 replies
Toviaz reviews and how it works
Hi. I'm having a little urinary incontinence trouble and would like to find out how Toviaz (fesoterodine fumarate) w... 1 reply
Hi. I'm having a little urinary incontinence trouble and would like to find out how Toviaz (fesoterodine fumarate) w... 1 reply
Toviaz has stopped working
I have been taking Toviaz for several years with great results. Recently it has stopped working and my old symptoms have...
I have been taking Toviaz for several years with great results. Recently it has stopped working and my old symptoms have...
Toviaz and Low Cost Alternatives
Dr. gave samples of Toviaz to husband for his urinary urgency problem, now husband wants to fill a prescription and afte...
Dr. gave samples of Toviaz to husband for his urinary urgency problem, now husband wants to fill a prescription and afte...
Ataxia And Toviaz Side Effects
CAN TOVIAZ TAKEN FOR A FEW YEARS MIMIC SYMPTOMS OF ATAXIA? i AM A PETITE WOMEN OF 95 LBS - 125 LBS (DIFFERENCES DUE TO S... 1 reply
CAN TOVIAZ TAKEN FOR A FEW YEARS MIMIC SYMPTOMS OF ATAXIA? i AM A PETITE WOMEN OF 95 LBS - 125 LBS (DIFFERENCES DUE TO S... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.