0378-7096 : Bromocriptine 5 mg (Bromocriptine Mesylate 5.74 mg) Oral Capsule
| NDC: | 0378-7096 |
| Labeler: | Mylan Pharmaceuticals Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Bromocriptine Mesylate Bromocriptine Mesylate |
| Dosage Form: | Oral Capsule |
| Application #: | ANDA077226 |
| Rev. Date: |
Appearance:
| Markings: | MYLAN;7096 |
| Shapes: |
Capsule |
| Colors: |
 Brown / Brown /
 White White |
| Size (mm): | 14 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
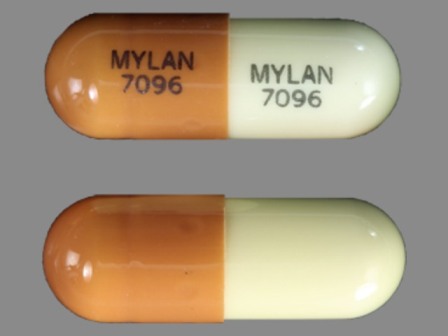
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 0378-7096-01: 100 CAPSULE IN 1 BOTTLE, UNIT‑DOSE (0378‑7096‑01)
- 0378-7096-93: 30 CAPSULE IN 1 BOTTLE, UNIT‑DOSE (0378‑7096‑93)
Active Ingredients:
- Bromocriptine Mesylate
Dosage Strength:
- 5 mg
Inactive Ingredients:
- Anhydrous Lactose
- Ferrosoferric Oxide
- D&c Yellow No. 10
- Fd&c Blue No. 1
- Fd&c Blue No. 2
- Fd&c Red No. 40
- Gelatin
- Magnesium Stearate
- Maleic Acid
- Butyl Alcohol
- Starch, Corn
- Propylene Glycol
- Ferric Oxide Red
- Shellac
- Sodium Lauryl Sulfate
- Titanium Dioxide
- Ferric Oxide Yellow
Pharmaceutical Classes:
- Ergolines [Chemical/Ingredient]
- Ergot Derivative [EPC]
Related Products:
Based on records with the same trade name.- 0378-2042 Bromocriptine (As Bromocriptine Mesylate) 2.5 mg Oral Tablet by Mylan Pharmaceuticals Inc.
- 0179-0165 Bromocriptine Mesylate 2.5 mg Oral Tablet by Kaiser Foundation Hospitals
- 0179-0225 Bromocriptine Mesylate 2.5 mg Oral Tablet by Kaiser Foundation Hospitals
- 0574-0106 Bromocriptine (As Bromocriptine Mesylate) 2.5 mg Oral Tablet by Paddock Laboratories, LLC
- 0781-2119 Bromocriptine 5 mg (Bromocriptine Mesylate 5.74 mg) Oral Capsule by Sandoz Inc
- 0781-5325 Bromocriptine (As Bromocriptine Mesylate) 2.5 mg Oral Tablet by Sandoz Inc
- 54868-5667 Bromocriptine (As Bromocriptine Mesylate) 2.5 mg Oral Tablet by Physicians Total Care, Inc.
- 60687-286 Bromocriptine Mesylate 2.5 mg Oral Tablet by American Health Packaging
- 63304-158 Bromocriptine Mesylate 5 mg Oral Capsule by Ranbaxy Pharmaceuticals Inc.
- 63304-962 Bromocriptine Mesylate 2.5 mg Oral Tablet by Ranbaxy Pharmaceuticals Inc.
- 65841-654 Bromocriptine 5 mg (Bromocriptine Mesylate 5.74 mg) Oral Capsule by Cadila Healthcare Limited
- 66685-5905 Bromocriptine (As Bromocriptine Mesylate) 2.5 mg Oral Tablet by Lek Pharmaceuticals
- 68084-375 Bromocriptine 5 mg (Bromocriptine Mesylate 5.74 mg) Oral Capsule by American Health Packaging
- 68151-1305 Bromocriptine Mesylate 2.5 mg Oral Tablet by Carilion Materials Management
- 68382-110 Bromocriptine 5 mg (Bromocriptine Mesylate 5.74 mg) Oral Capsule by Zydus Pharmaceuticals (Usa) Inc.
- 69189-7096 Bromocriptine Mesylate 5 mg Oral Capsule by Avera Mckennan Hospital
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 0378-7077Next: 0378-7097 >
Related Discussions:
Bromocriptine usage and dodage
i was prescribed with Bromocriptine, but i'am confused beacuse i see there are two types: the capsules which are 5mg... 2 replies
i was prescribed with Bromocriptine, but i'am confused beacuse i see there are two types: the capsules which are 5mg... 2 replies
Bromocriptine MesylateTab 2.5 MG
The other name for this Prescription Drug is Paredell. ## Thank you for submitting this medicine. You can find more info... 1 reply
The other name for this Prescription Drug is Paredell. ## Thank you for submitting this medicine. You can find more info... 1 reply
bromocriptine
indications, side effects of this drug...
indications, side effects of this drug...
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.