0068-0510 : Rifadin 150 mg Oral Capsule
| NDC: | 0068-0510 |
| Labeler: | Sanofi-aventis U.S. LLC |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Rifadin Rifadin |
| Dosage Form: | Oral Capsule |
| Application #: | ANDA062303 |
| Rev. Date: |
Appearance:
| Markings: | RIFADIN;150 |
| Shapes: |
Capsule |
| Colors: |
 Red Red |
| Size (mm): | 14 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
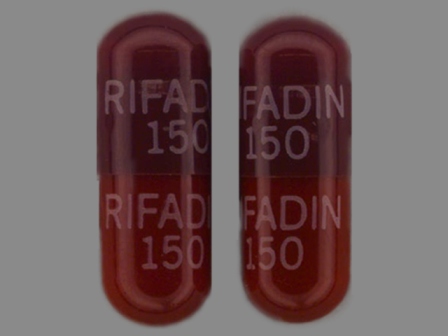
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 0068-0510-30: 30 CAPSULE IN 1 BOTTLE (0068‑0510‑30)
Active Ingredients:
- Rifampin
Dosage Strength:
- 150 mg
Inactive Ingredients:
- Starch, Corn
- D&c Red No. 28
- Fd&c Blue No. 1
- Fd&c Red No. 40
- Gelatin
- Magnesium Stearate
- Titanium Dioxide
Pharmaceutical Classes:
- Rifamycin Antibacterial [EPC]
- Rifamycins [Chemical/Ingredient]
Related Products:
Based on records with the same trade name.- 0068-0508 Rifadin 300 mg Oral Capsule by Sanofi-aventis U.S. LLC
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 0068-0509Next: 0068-0597 >
Related Discussions:
R Cinex Rifampin And Isoniazid Capsule U P S
I have been taking R Cinex Rifampin And Isoniazid Capsule U P S since last 67 days but I missed 1day dose,which doctor p... 3 replies
I have been taking R Cinex Rifampin And Isoniazid Capsule U P S since last 67 days but I missed 1day dose,which doctor p... 3 replies
Side effects of Rifampin (rifampicin)
Hello, I would like to know if anyone is taking Rifampin and what are side effects, i read about it online but would lik... 1 reply
Hello, I would like to know if anyone is taking Rifampin and what are side effects, i read about it online but would lik... 1 reply
rifampin 150mg tablet availability
I need to locate the above mentioned medicine. I can not locate this drug in tablet form locally. Can anyone tell me abo...
I need to locate the above mentioned medicine. I can not locate this drug in tablet form locally. Can anyone tell me abo...
rifampin
infection...
infection...
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.