0056-0521 : Sinemet CR 50-200 Extended Release Tablet
| NDC: | 0056-0521 |
| Labeler: | Bristol-myers Squibb Pharma Company |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Sinemet CR Sinemet CR |
| Dosage Form: | Oral Tablet, Extended Release |
| Application #: | NDA019856 |
| Rev. Date: |
Appearance:
| Markings: | 521;SINEMET;CR |
| Shapes: |
Oval |
| Colors: |
 Orange Orange |
| Size (mm): | 13 |
| Segments: * | 2 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 2 indicates a scored pill which can be broken into 2 equal pieces. | |
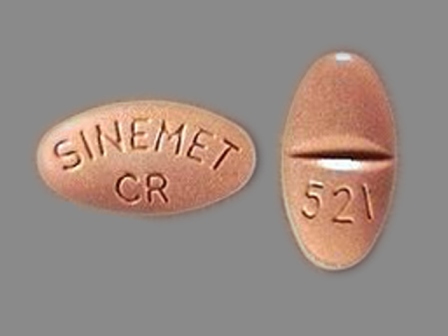
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 0056-0521-68: 100 TABLET, EXTENDED RELEASE IN 1 BOTTLE (0056‑0521‑68)
- 0056-0521-85: 500 TABLET, EXTENDED RELEASE IN 1 BOTTLE (0056‑0521‑85)
Active Ingredients:
- Carbidopa
- Levodopa
Dosage Strength:
- 50 mg
- 200 mg
Inactive Ingredients:
- Hydroxypropyl Cellulose
- Magnesium Stearate
- Ferric Oxide Red
- D&c Yellow No. 10
Pharmaceutical Classes:
- Aromatic Amino Acid Decarboxylation Inhibitor [EPC]
- DOPA Decarboxylase Inhibitors [MoA]
Related Products:
Based on records with the same trade name.- 0056-0601 Sinemet CR (Carbidopa 25 mg / Levodopa 100 mg) Extended Release Tablet by Bristol-myers Squibb Pharma Company
- 0056-0647 Sinemet 10/100 Oral Tablet by Bristol-myers Squibb Pharma Company
- 0056-0650 Sinemet 25-100 Oral Tablet by Bristol-myers Squibb Pharma Company
- 0056-0654 Sinemet 25/250 Oral Tablet by Bristol-myers Squibb Pharma Company
- 0006-3915 Sinemet 10/100 Oral Tablet by Merck Sharp & Dohme Corp.
- 0006-3916 Sinemet 25-100 Oral Tablet by Merck Sharp & Dohme Corp.
- 0006-3917 Sinemet 25/250 Oral Tablet by Merck Sharp & Dohme Corp.
- 0006-3918 Sinemet CR (Carbidopa 25 mg / Levodopa 100 mg) Extended Release Tablet by Merck Sharp & Dohme Corp.
- 0006-3919 Sinemet CR 50-200 Extended Release Tablet by Merck Sharp & Dohme Corp.
- 0006-6722 Sinemet Oral Tablet by Merck Sharp & Dohme Corp.
- 0006-6723 Sinemet Oral Tablet by Merck Sharp & Dohme Corp.
- 0006-6724 Sinemet Oral Tablet by Merck Sharp & Dohme Corp.
- 78206-166 Sinemet Oral Tablet by Organon LLC
- 78206-167 Sinemet Oral Tablet by Organon LLC
- 78206-168 Sinemet Oral Tablet by Organon LLC
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 0056-0510Next: 0056-0601 >
Related Discussions:
Carbidopa/Levodopa Drug Information
Parkinsons disease ## would like to know all side effects for this medication ## 3rd day on carb/levo, feeling sick, sle... 7 replies
Parkinsons disease ## would like to know all side effects for this medication ## 3rd day on carb/levo, feeling sick, sle... 7 replies
Carbidopa Levodopa 93 / 293
Round yellow scored tablet 93/293 ## My mother-in-law takes carbidopa (sinemet). Part of her pills say 93-293, and part ... 4 replies
Round yellow scored tablet 93/293 ## My mother-in-law takes carbidopa (sinemet). Part of her pills say 93-293, and part ... 4 replies
Carbidopa Levodopa With Food Or Antacid
Do I have to give this to my mom before food or an antacid pill? ## Hello, Robert! How are you? This medication should b... 1 reply
Do I have to give this to my mom before food or an antacid pill? ## Hello, Robert! How are you? This medication should b... 1 reply
Carbidopa Levodopa ER
what is the color? any numbers? ## Once a medication is available in generic form, it is manufactured by many different ... 2 replies
what is the color? any numbers? ## Once a medication is available in generic form, it is manufactured by many different ... 2 replies
levodopa and carbidopa
What are the risks of combining these two drugs with LUMIGAN, ELIPTIC AND DUOTRAN (prescribed for glaucoma)? ## Hello, C... 1 reply
What are the risks of combining these two drugs with LUMIGAN, ELIPTIC AND DUOTRAN (prescribed for glaucoma)? ## Hello, C... 1 reply
generic Rytary - carbidopa and levodopa
Is this medication available in a generic form yet, or can I only get my prescription filled with the brand name? ## The... 1 reply
Is this medication available in a generic form yet, or can I only get my prescription filled with the brand name? ## The... 1 reply
carbidopa 25/levadopa 100 mg tab and carbidopa 25/ levodopa 100mg sa tab
what is the difference? between carbidopa 25/levodopa 100mg tab and carbidopa 25/levodopa 100mg sa tab ## Hello, Dave! H... 1 reply
what is the difference? between carbidopa 25/levodopa 100mg tab and carbidopa 25/levodopa 100mg sa tab ## Hello, Dave! H... 1 reply
difficulty facing with levodopa and carbidopa
My wife is suffering from PD in the age of 36 year and the doctor was advised syndopa plus, but after three years she ha... 2 replies
My wife is suffering from PD in the age of 36 year and the doctor was advised syndopa plus, but after three years she ha... 2 replies
carbidopa/levo/ebt cap 50/200/200
Are there any programs that can offer assistance with my monthly medications? ## Hello, Dale! How are you? Each page of ... 1 reply
Are there any programs that can offer assistance with my monthly medications? ## Hello, Dale! How are you? Each page of ... 1 reply
levadopa/carbidopa
my seven-year-old has symptoms of dystonia and his geneticist is recommending we try putting him on a dopa trial--i am n...
my seven-year-old has symptoms of dystonia and his geneticist is recommending we try putting him on a dopa trial--i am n...
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.