Entocort Ec
10 Topics Found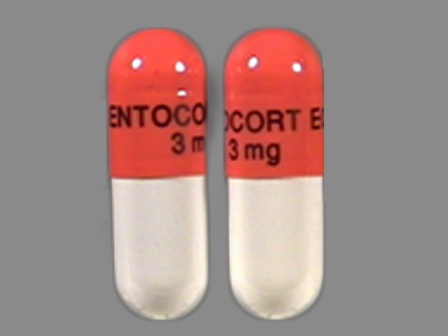
- NDC: *
- 0186-0702
- Imprint:
- ENTOCORT EC 3 mg
- Color:
- Gray
- Shape:
- Capsule
- Size:
- 7 mm
- Segments:
- 1
- Drug:
- Entocort EC
- Labeler: *
- Astrazeneca Lp
- Category:
- Anti-Inflammatory
Source: US National Library of Medicine (NLM)
Budesonide (BUD), sold under the brand name Pulmicort among others, is a medication of the corticosteroid type.[3] It is available as an inhaler, nebulization solution, pill, nasal spray, and rectal forms.[3][4] The inhaled form is used in the long-term management of asthma and chronic obstructive pulmonary disease (COPD).[3][5][6] The nasal spray is used for allergic rhinitis and nasal polyps.&...
I have been gaining weight on this medication. Is it better than Predizone for you? ## It is in the same drug class, so the side effects are the same, including weight gain. ## is there canadian outlet for entocort ec prefferably mail order?
Is there a Patient Assistant Program for Entocort EC that can help me get my medication I can not afford? ## Hello, Mary! How are you? I'm sorry that you cannot afford your medication. If you have insurance, you may be able to get help paying the rest of the cost from the Patient Access Network Foundation, it's what they specialize in, to ensure that people can get the medications they need. To see if you qualify, you can call them for more information at 866-316-7263. Alternatively, towards the lower right of each page on this site, there is a printable pharmacy discount card that you use, it is free and may save you money on the prescription. As listed by the FDA, Entocort EC is used to treat mild to moderate Crohn's Disease. Its typical side effects are listed as possibly...
I have been on entocort off and on since 2012 due to gastro diverticulitis (highly flammable). After a colonopscopy in Sept. 2012 I was given entocort for 3 a day-1 mo., then 2 a day-1 mo., then 1 a day-1 mo. Then on Feb. 7th, 2013 I fell on cement and was taken to a hospital by helicopter. They kept me alive due to help from the Dr.'s at University of Ky. Hospital -Lexington, Ky. Then later they gave me prescription budesonide ec 3mg. 1 per day. After another colonopscopy on 9/24/17 they ruled it as diverticulitis w/colitis again. Recently they changed my script to budesonide 3mg with the drug co. listed as MYLAN 7155- NO EC letters listed on the script. My question is: Is this sufficient or as good as the ENTOCORT EC 3-mg. in your opinion? This is my question about this prescripti...
Entocort Ec 3 mg Oral Capsule by Paddock Laboratories LLC ## Markings: ENTOCORT;EC;3mg ## Shape: Capsule ## Color: Gray ## Package Codes: 0574-9850-10 ## Active Ingredients: Budesonide
24 Hr Entocort 3 mg Extended Release Enteric Coated Capsule by Astrazeneca Lp ## Markings: ENTOCORT;EC;3;mg ## Shape: Capsule ## Color: Gray ## Package Codes: 0186-0702-10, 0186-0702-94 ## Active Ingredients: Budesonide
24 Hr Entocort 3 mg Extended Release Enteric Coated Capsule by Prometheus Laboratories Inc ## Markings: ENTOCORT;EC;3;mg ## Shape: Capsule ## Color: Gray ## Package Codes: 65483-702-10 ## Active Ingredients: Budesonide
24 Hr Entocort 3 mg Extended Release Enteric Coated Capsule by Physicians Total Care, Inc. ## Markings: ENTOCORT;EC;3;mg ## Shape: Capsule ## Color: Gray ## Package Codes: 54868-4910-0, 54868-4910-1, 54868-4910-2, 54868-4910-3 ## Active Ingredients: Budesonide
Budesonide 3 mg Oral Capsule by Paddock Laboratories, LLC ## Markings: ENTOCORT;EC;3mg ## Shape: Capsule ## Color: Gray ## Package Codes: 0574-9855-10