Fenofibrate - NDC Database (Page 3)
383 records found
Start a Discussion
35356-905 Feb 01, 2010
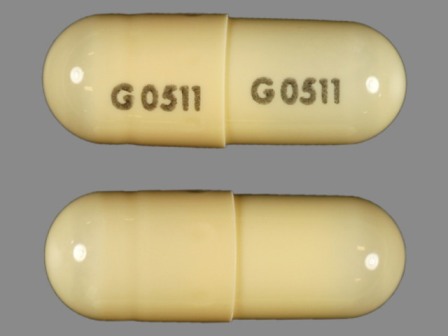
Fenofibrate 134 mg Oral Capsule by Lake Erie Medical Dba Quality Care Products LLC
35356-916 Feb 01, 2010
Fenofibrate 160 mg Oral Tablet by Lake Erie Medical Dba Quality Care Products LLC
35356-925 Feb 01, 2010
Fenofibrate 67 mg Oral Capsule by Lake Erie Medical Dba Quality Care Products LLC
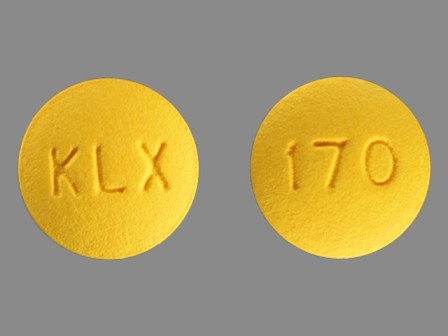
42043-170 Nov 20, 2009
Fenofibrate 54 mg Oral Tablet by Karalex Pharma LLC
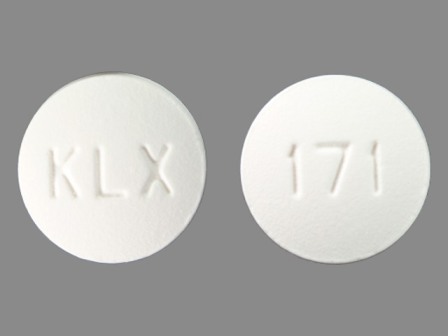
42043-171 Nov 20, 2009
Fenofibrate 160 mg Oral Tablet by Karalex Pharma LLC
42291-289 Apr 01, 2015
Fenofibrate 54 mg Oral Tablet by Avkare, Inc.
42291-290 Jan 16, 2015
Fenofibrate 160 mg Oral Tablet by Avkare, Inc.
43353-001 May 10, 2006
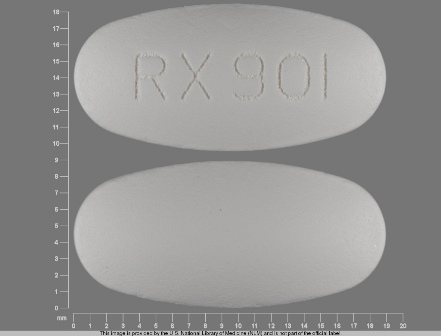
Fenofibrate 160 mg Oral Tablet, Film Coated by Aphena Pharma Solutions - Tennessee, LLC
43353-248 Aug 13, 2014
Fenofibrate 48 mg Oral Tablet by Aphena Pharma Solutions - Tennessee, LLC
43353-898 Dec 22, 2009
Fenofibrate 160 mg Oral Tablet, Film Coated by Aphena Pharma Solutions - Tennessee, LLC
45802-132 Sep 23, 2014
Fenofibrate 145 mg Oral Tablet by Perrigo New York Inc
50090-1275 Sep 18, 2020
Fenofibrate 160 mg Oral Tablet, Film Coated by A-s Medication Solutions
50090-2112 Nov 05, 2004
Fenofibrate 48 mg Oral Tablet by A-s Medication Solutions
50090-2459 Sep 23, 2014
Fenofibrate 145 mg Oral Tablet by A-s Medication Solutions
50090-2692 Aug 13, 2014
Fenofibrate 48 mg Oral Tablet by A-s Medication Solutions
50090-2857 Feb 09, 2016
Fenofibrate 145 mg Oral Tablet by A-s Medication Solutions
50090-3255 Jul 02, 2018
Fenofibrate 54 mg Oral Tablet, Film Coated by A-s Medication Solutions
50090-3904 Nov 19, 2012
Fenofibrate 145 mg Oral Tablet by A-s Medication Solutions
50268-309 Nov 02, 2020
Fenofibrate 134 mg Oral Capsule by Avpak
50268-310 Sep 19, 2016
Fenofibrate 48 mg Oral Tablet by Avpak
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us .