63481-025 : Frova 2.5 mg Oral Tablet
| NDC: | 63481-025 |
| Labeler: | Endo Pharmaceuticals Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Frova Frova |
| Dosage Form: | Oral Tablet, Film Coated |
| Application #: | NDA021006 |
| Rev. Date: |
Appearance:
| Markings: | E;2;5 |
| Shapes: |
Round |
| Colors: |
 White White |
| Size (mm): | 8 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
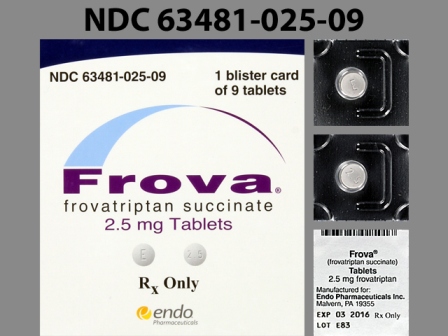
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 63481-025-09: 1 BLISTER PACK IN 1 CARTON (63481‑025‑09) > 9 TABLET, FILM COATED IN 1 BLISTER PACK
- 63481-025-12: 6 BLISTER PACK IN 1 CARTON (63481‑025‑12) > 2 TABLET, FILM COATED IN 1 BLISTER PACK
Active Ingredients:
- Frovatriptan Succinate
Dosage Strength:
- 2.5 mg
Inactive Ingredients:
- Lactose
- Cellulose, Microcrystalline
- Silicon Dioxide
- Sodium Starch Glycolate Type a Potato
- Magnesium Stearate
- Hypromelloses
- Polyethylene Glycol 3000
- Triacetin
- Titanium Dioxide
Pharmaceutical Classes:
- Serotonin 1b Receptor Agonists [MoA]
- Serotonin 1d Receptor Agonists [MoA]
- Serotonin-1b and Serotonin-1d Receptor Agonist [EPC]
Related Products:
Based on records with the same trade name.- 16590-396 Frova 2.5 mg Oral Tablet by Stat Rx USA LLC
- 35356-169 Frova 2.5 mg Oral Tablet by Lake Erie Medical Dba Quality Care Products LLC
- 50436-0026 Frova 2.5 mg Oral Tablet, Film Coated by Unit Dose Services
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 63479-2556Next: 63481-042 >
Related Discussions:
Metoprolol Succinate ER Causes Hair Loss
Why do doctors online and offline pretend that Metropolol does NOT cause hair loss, when there are patients online and r... 15 replies
Why do doctors online and offline pretend that Metropolol does NOT cause hair loss, when there are patients online and r... 15 replies
metoprolol succinate er 50mg tabs
medium size, white, oval, scored tablet, number 369 imprinted on tablet ## Metoprolol Succinate Er 50mg Tabs with number... 9 replies
medium size, white, oval, scored tablet, number 369 imprinted on tablet ## Metoprolol Succinate Er 50mg Tabs with number... 9 replies
metoprolol succinate
25 mg extended release tablets ## wouldl like to know the side affects ## I was prescribed 25mg of Metoprolol Succinate ... 5 replies
25 mg extended release tablets ## wouldl like to know the side affects ## I was prescribed 25mg of Metoprolol Succinate ... 5 replies
metoprolol succinate er tab 50mg
I would like to know why I have this drug metoprolol succinate ER 25 if it was recalled. Is it now considerred safe? Has... 4 replies
I would like to know why I have this drug metoprolol succinate ER 25 if it was recalled. Is it now considerred safe? Has... 4 replies
Metoprolol Succinate ER 100mg
Metoprolol Succunate ER 100mg scored, white, round tablet with 100 over m imprinted on the back ## Does it make any diff... 4 replies
Metoprolol Succunate ER 100mg scored, white, round tablet with 100 over m imprinted on the back ## Does it make any diff... 4 replies
Metoprolol Succinate ER
I switched pharmacies and the pills look different - it didn't concern me, and research says they are the same thing... 3 replies
I switched pharmacies and the pills look different - it didn't concern me, and research says they are the same thing... 3 replies
metoprolol succinate extended release tablets
C 75 Light Blue 100 Mg. ## I have order metoprolol succinate extended release capsules 50mg from Canada Pram. I have a d... 3 replies
C 75 Light Blue 100 Mg. ## I have order metoprolol succinate extended release capsules 50mg from Canada Pram. I have a d... 3 replies
metoprolol succinate 50MG SA Tab
Does SA mean that this is an extended release pill ???? ## Means Sustained Release. ## Yes, SA stands for sustained acti... 2 replies
Does SA mean that this is an extended release pill ???? ## Means Sustained Release. ## Yes, SA stands for sustained acti... 2 replies
Metoprolol Succinate - Discontinuing - Wean or not?
I've been on 25mg, Extended Release, of Metroprolol Succinate for a while, about four or five years I think - never ... 2 replies
I've been on 25mg, Extended Release, of Metroprolol Succinate for a while, about four or five years I think - never ... 2 replies
Metoprolol succinate 25mg
I'm so tired... have been taking it for 1 month and getting more tired each day. Is this a side effect? ## Have you ... 2 replies
I'm so tired... have been taking it for 1 month and getting more tired each day. Is this a side effect? ## Have you ... 2 replies
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.