55154-3122 : Cellcept 250 mg Oral Capsule
| NDC: | 55154-3122 |
| Labeler: | Cardinal Health |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Cellcept Cellcept |
| Dosage Form: | Oral Capsule |
| Application #: | NDA050722 |
| Rev. Date: |
Appearance:
| Markings: | CellCept;250;Roche |
| Shapes: |
Capsule |
| Colors: |
 Blue / Blue /
 Brown Brown |
| Size (mm): | 19 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
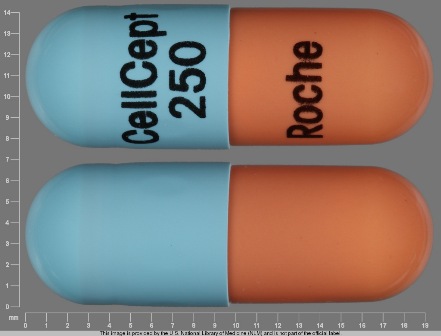
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 55154-3122-0: 10 POUCH IN 1 BAG (55154‑3122‑0) > 1 CAPSULE IN 1 POUCH
- 55154-3122-4: 100 POUCH IN 1 CARTON (55154‑3122‑4) > 1 CAPSULE IN 1 POUCH (55154‑3122‑6)
Active Ingredients:
- Mycophenolate Mofetil
Dosage Strength:
- 250 mg
Inactive Ingredients:
- Croscarmellose Sodium
- Magnesium Stearate
- Povidone K90
- Starch, Corn
- Ferrosoferric Oxide
- Fd&c Blue No. 2
- Gelatin
- Ferric Oxide Yellow
- Ferric Oxide Red
- Titanium Dioxide
- Silicon Dioxide
- Sodium Lauryl Sulfate
Pharmaceutical Classes:
- Antimetabolite Immunosuppressant [EPC]
Related Products:
Based on records with the same trade name.- 0004-0259 Cellcept 250 mg Oral Capsule by Genentech, Inc.
- 0004-0260 Cellcept 500 mg Oral Tablet by Genentech, Inc.
- 0004-0261 Cellcept 200 mg/ml Oral Powder, for Suspension by Genentech, Inc.
- 0004-0298 Cellcept 500 mg/20ml Intravenous Injection, Powder, Lyophilized, for Solution by Genentech, Inc.
- 21695-171 Cellcept 250 mg Oral Capsule by Rebel Distributors Corp
- 49999-936 Cellcept 250 mg Oral Capsule by Lake Erie Medical & Surgical Supply Dba Quality Care Products LLC
- 49999-937 Cellcept 500 mg Oral Tablet by Lake Erie Medical & Surgical Supply Dba Quality Care Products LLC
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 55154-3117Next: 55154-3182 >
Related Discussions:
Pomegranate interactions with Tacrolimus and Mycophenolate
Can I take a tonic that includes pomegranate supplement when taking Tacrolimus and Mycophenolate anti-kidney-rejection m... 1 reply
Can I take a tonic that includes pomegranate supplement when taking Tacrolimus and Mycophenolate anti-kidney-rejection m... 1 reply
cellcept for multiple sclerosis
cellcept was prescribed for me by neurologist for multiple sclerosis. Have had MS for 20 years and had symptom progressi... 62 replies
cellcept was prescribed for me by neurologist for multiple sclerosis. Have had MS for 20 years and had symptom progressi... 62 replies
Cellcept for CIDP
I have started taking Cellcept for the treatment of CIDP. I am interested in knowing if anyone else is taking cellcept f... 14 replies
I have started taking Cellcept for the treatment of CIDP. I am interested in knowing if anyone else is taking cellcept f... 14 replies
cellcept expectations?
I have been on cellcept for many years for severe systemic lupus. I am happy to say that I am in remission and have aece... 4 replies
I have been on cellcept for many years for severe systemic lupus. I am happy to say that I am in remission and have aece... 4 replies
Cellcept - when I can eat popsicles
I started cellcept yesterday for a liver problem. It says I can't eat 2 hours before and 4 hours after. I would like... 4 replies
I started cellcept yesterday for a liver problem. It says I can't eat 2 hours before and 4 hours after. I would like... 4 replies
Cellcept meals
How long before and after meal should i wait to take cellcept? I don't want to take to soon.I've been on it 5 da... 1 reply
How long before and after meal should i wait to take cellcept? I don't want to take to soon.I've been on it 5 da... 1 reply
Cellcept pregnancy
Hi, my name is lucy. I am 25 years old. I had a liver transplant about 2 years ago. I am currently taking cellcept. I am...
Hi, my name is lucy. I am 25 years old. I had a liver transplant about 2 years ago. I am currently taking cellcept. I am...
new to cellcept
I am starting cellcept tomorrow so that I can finally go off of imuran and eventually prednisone. Have been on many meds... 10 replies
I am starting cellcept tomorrow so that I can finally go off of imuran and eventually prednisone. Have been on many meds... 10 replies
Reduced cellcept dose from 2000/day to 1000/day
My doctor has approved a drop from 2000/day to 1000/day and I am experiencing a fogginess and lack of pep. I use it with... 1 reply
My doctor has approved a drop from 2000/day to 1000/day and I am experiencing a fogginess and lack of pep. I use it with... 1 reply
Forskolin, Atenolol, Amlodipine, Tacrolimus, Bactrim, Humalog & Cellcept
I had a kidney transplant and am on the following medications: Tacrolimus, cellcept, amlodipine, atenolol, bactrim &... 1 reply
I had a kidney transplant and am on the following medications: Tacrolimus, cellcept, amlodipine, atenolol, bactrim &... 1 reply
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.