54868-5945 : Acarbose 25 mg Oral Tablet
| NDC: | 54868-5945 |
| Labeler: | Physicians Total Care, Inc. |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Acarbose Acarbose |
| Dosage Form: | Oral Tablet |
| Application #: | ANDA078470 |
| Rev. Date: |
Appearance:
| Markings: | 54;311 |
| Shapes: |
Round |
| Colors: |
 White White |
| Size (mm): | 6 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
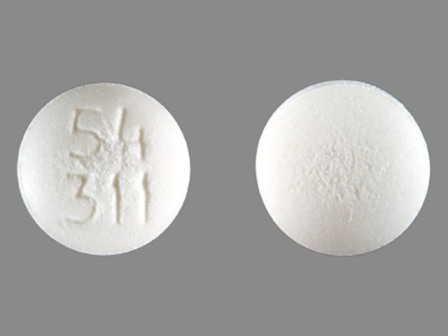
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 54868-5945-0: 90 TABLET IN 1 BOTTLE, PLASTIC (54868‑5945‑0)
Active Ingredients:
- Acarbose
Dosage Strength:
- 25 mg
Inactive Ingredients:
- Silicon Dioxide
- Starch, Corn
- Magnesium Stearate
- Cellulose, Microcrystalline
Pharmaceutical Classes:
- alpha Glucosidase Inhibitors [MoA]
- alpha-Glucosidase Inhibitor [EPC]
Related Products:
Based on records with the same trade name.- 0054-0140 Acarbose 25 mg Oral Tablet by Roxane Laboratories, Inc
- 0054-0141 Acarbose 50 mg Oral Tablet by Roxane Laboratories, Inc
- 0054-0142 Acarbose 100 mg Oral Tablet by Roxane Laboratories, Inc
- 0115-1150 Acarbose 25 mg Oral Tablet by Global Pharmaceuticals, Division of Impax Laboratories, Inc.
- 0115-1151 Acarbose 50 mg Oral Tablet by Global Pharmaceuticals, Division of Impax Laboratories, Inc.
- 0115-1152 Acarbose 100 mg Oral Tablet by Global Pharmaceuticals, Division of Impax Laboratories, Inc.
- 0378-2820 Acarbose 25 mg Oral Tablet by Mylan Pharmaceuticals Inc.
- 0378-2821 Acarbose 50 mg Oral Tablet by Mylan Pharmaceuticals Inc.
- 0378-2822 Acarbose 100 mg Oral Tablet by Mylan Pharmaceuticals Inc.
- 11788-039 Acarbose 25 mg Oral Tablet by Aiping Pharmaceutical
- 11788-040 Acarbose 50 mg Oral Tablet by Aiping Pharmaceutical
- 11788-041 Acarbose 100 mg Oral Tablet by Aiping Pharmaceutical
- 16252-523 Acarbose 25 mg Oral Tablet by Cobalt Laboratories
- 16252-524 Acarbose 50 mg Oral Tablet by Cobalt Laboratories
- 16252-525 Acarbose 100 mg Oral Tablet by Cobalt Laboratories
- 23155-147 Acarbose 25 mg Oral Tablet by Heritage Pharmaceuticals Inc.
- 23155-148 Acarbose 50 mg Oral Tablet by Heritage Pharmaceuticals Inc.
- 23155-149 Acarbose 100 mg Oral Tablet by Heritage Pharmaceuticals Inc.
- 42291-130 Acarbose 25 mg/1 Oral Tablet by Avkare, Inc.
- 42291-131 Acarbose 50 mg/1 Oral Tablet by Avkare, Inc.
- More related products ...
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 54868-5944Next: 54868-5950 >
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.