50090-1307 : Bystolic 10 mg Oral Tablet
| NDC: | 50090-1307 |
| Labeler: | A-s Medication Solutions |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Bystolic Bystolic |
| Dosage Form: | Oral Tablet |
| Application #: | NDA021742 |
| Rev. Date: |
Appearance:
| Markings: | 10;FL |
| Shapes: |
Triangle (3 sides) |
| Colors: |
 Purple Purple |
| Size (mm): | 10 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
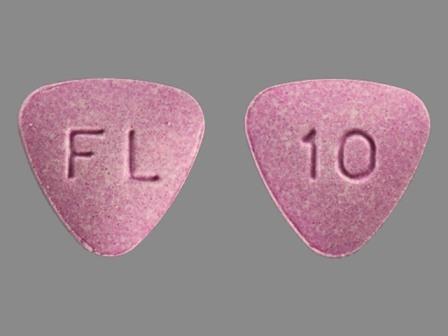
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 50090-1307-0: 30 TABLET IN 1 BOTTLE (50090‑1307‑0)
- 50090-1307-1: 90 TABLET IN 1 BOTTLE (50090‑1307‑1)
Active Ingredients:
- Nebivolol Hydrochloride
Dosage Strength:
- 10 mg
Inactive Ingredients:
- Silicon Dioxide
- Croscarmellose Sodium
- D&c Red No. 27
- Fd&c Blue No. 2
- Fd&c Yellow No. 6
- Hypromellose, Unspecified
- Lactose Monohydrate
- Magnesium Stearate
- Microcrystalline Cellulose
- Starch, Corn
- Polysorbate 80
- Sodium Lauryl Sulfate
Pharmaceutical Classes:
- Adrenergic beta-Antagonists [MoA]
- beta-Adrenergic Blocker [EPC]
Related Products:
Based on records with the same trade name.- 50090-1127 Bystolic 5 mg Oral Tablet by A-s Medication Solutions
- 50090-3326 Bystolic 20 mg Oral Tablet by A-s Medication Solutions
- 54569-6119 Bystolic 5 mg Oral Tablet by A-s Medication Solutions
- 0456-1402 Bystolic 2.5 mg Oral Tablet by Forest Laboratories, Inc.
- 0456-1405 Bystolic 5 mg Oral Tablet by Forest Laboratories, Inc.
- 0456-1410 Bystolic 10 mg Oral Tablet by Forest Laboratories, Inc.
- 0456-1420 Bystolic 20 mg Oral Tablet by Forest Laboratories, Inc.
- 54868-5944 Bystolic 5 mg Oral Tablet by Physicians Total Care, Inc.
- 54868-6018 Bystolic 10 mg Oral Tablet by Physicians Total Care, Inc.
- 55154-4621 Bystolic 5 mg Oral Tablet by Cardinal Health
- 55154-4622 Bystolic 20 mg Oral Tablet by Cardinal Health
- 55154-4623 Bystolic 10 mg Oral Tablet by Cardinal Health
- 68151-5135 Bystolic 10 mg Oral Tablet by Carilion Materials Management
- 69189-1405 Bystolic 5 mg Oral Tablet by Avera Mckennan Hospital
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 50090-1306Next: 50090-1308 >
Related Discussions:
Nebivolol and excessive head sweating
I took Bystolic when I was first diagnosed with high blood pressure and did fine. When the generic came out I was switch... 2 replies
I took Bystolic when I was first diagnosed with high blood pressure and did fine. When the generic came out I was switch... 2 replies
Nebivolol, Olmesartan, Amlodipine and Viagra
I am 42 years old taking nebivolol 5mg, olmesartan medoxomil 40 mg and amlodipine 5 mg for last 10 years for my blood pr... 1 reply
I am 42 years old taking nebivolol 5mg, olmesartan medoxomil 40 mg and amlodipine 5 mg for last 10 years for my blood pr... 1 reply
Nebivolol stoppage after long term usage
I have been taking Nebivolol 2.5 mg for 10 years to control hypertension. Can it cause cough which is persisting despite...
I have been taking Nebivolol 2.5 mg for 10 years to control hypertension. Can it cause cough which is persisting despite...
Viagra and Nebivolol?
I am taking 1.5 mg of Nebivolol daily. I am suspicious that Viagra could be a problem with potentiation. Anyone know? ##... 3 replies
I am taking 1.5 mg of Nebivolol daily. I am suspicious that Viagra could be a problem with potentiation. Anyone know? ##... 3 replies
equivalent doses of nebivolol and atenolol
Can 25 mg of Atenolol be replaced by 5 mg or by 2.5mg of Nebivolol? ## Hello, Sharda! How are you? From what I've be... 1 reply
Can 25 mg of Atenolol be replaced by 5 mg or by 2.5mg of Nebivolol? ## Hello, Sharda! How are you? From what I've be... 1 reply
Bystolic 10mg for blood pressure
I recently went to fill my rx at my local pharmacy. I was told that bystolic would not be in until the end of the month ... 12 replies
I recently went to fill my rx at my local pharmacy. I was told that bystolic would not be in until the end of the month ... 12 replies
Bystolic left hand cold and pain
I am currently taking 5mg Bystolic. My left hand is cold off and on through out the day. During the cold hand, I also ex... 7 replies
I am currently taking 5mg Bystolic. My left hand is cold off and on through out the day. During the cold hand, I also ex... 7 replies
Bystolic and diiarrhea
Been taking Bystolic for three weeks, good BP results; however diarrhea has been an issue. Anyone else with this side ef... 6 replies
Been taking Bystolic for three weeks, good BP results; however diarrhea has been an issue. Anyone else with this side ef... 6 replies
BYSTOLIC: 5mg - Anyone Experience Stomach/Intestinal Issues?
I've been on this medication @ 2yrs after coming from Metoprolol 10mg. They were/are prescribed for lowering my hear... 5 replies
I've been on this medication @ 2yrs after coming from Metoprolol 10mg. They were/are prescribed for lowering my hear... 5 replies
Bystolic questions
Hi. I am taking a new beta blocker for 20 days now. I had to because of Sotalol and Apocard is not a good combination on... 5 replies
Hi. I am taking a new beta blocker for 20 days now. I had to because of Sotalol and Apocard is not a good combination on... 5 replies
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.