42708-027 : Hydrochlorothiazide 12.5 mg Oral Capsule, Gelatin Coated
| NDC: | 42708-027 |
| Labeler: | Qpharma Inc |
| Product Type: | Human Prescription Drug |
| Drug Name: |  Hydrochlorothiazide Hydrochlorothiazide |
| Dosage Form: | Oral Capsule, Gelatin Coated |
| Application #: | NDA020504 |
| Rev. Date: |
Appearance:
| Markings: | WATSON;347;and;12;5;mg |
| Shapes: |
Capsule |
| Colors: |
 White / White /
 Turquoise Turquoise |
| Size (mm): | 14 |
| Segments: * | 1 |
* Segments = the number of equally sized pieces which the pill can be broken into. In this case, a value of 1 indicates a solid pill with no score lines. | |
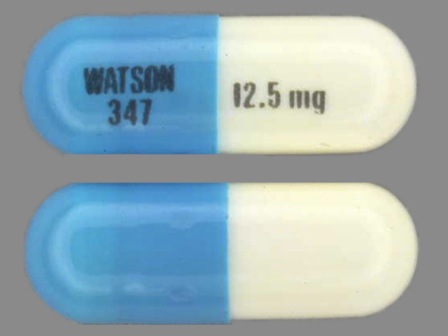
The above image is provided by the U.S. National Library of Medicine (NLM) and is not part of the official label.
NDC Package Codes:
- 42708-027-30: 30 CAPSULE, GELATIN COATED IN 1 BOTTLE (42708‑027‑30)
Active Ingredients:
- Hydrochlorothiazide
Dosage Strength:
- 12.5 mg
Inactive Ingredients:
- Silicon Dioxide
- Starch, Corn
- D&c Red No. 28
- D&c Yellow No. 10
- Fd&c Blue No. 1
- Gelatin, Unspecified
- Lactose Monohydrate
- Magnesium Stearate
- Titanium Dioxide
- Ferrosoferric Oxide
- Fd&c Blue No. 2
- Fd&c Red No. 40 /
Pharmaceutical Classes:
- Increased Diuresis [PE]
- Thiazide Diuretic [EPC]
- Thiazides [CS]
Related Products:
Based on records with the same trade name.- 42708-028 Hydrochlorothiazide 25 mg Oral Tablet by Qpharma Inc
- 42708-079 Hydrochlorothiazide 25 mg Oral Tablet by Qpharma Inc
- 42708-086 Hydrochlorothiazide 12.5 mg Oral Capsule by Qpharma Inc
- 42708-164 Hydrochlorothiazide 25 mg Oral Tablet by Qpharma, Inc.
- 0093-2080 Hctz 12.5 mg Oral Capsule by Teva Pharmaceuticals USA Inc
- 0143-1256 Hctz 25 mg Oral Tablet by West-ward Pharmaceutical Corp
- 0143-1257 Hctz 50 mg Oral Tablet by West-ward Pharmaceutical Corp
- 0143-3125 Hctz 12.5 mg Oral Capsule by West-ward Pharmaceutical Corp
- 0172-2083 Hctz 25 mg Oral Tablet by Ivax Pharmaceuticals, Inc.
- 0172-2089 Hctz 50 mg Oral Tablet by Ivax Pharmaceuticals, Inc.
- 0179-0065 Hctz 25 mg Oral Tablet by Kaiser Foundation Hospitals
- 0228-2221 Hctz 25 mg Oral Tablet by Actavis Elizabeth LLC
- 0228-2222 Hctz 50 mg Oral Tablet by Actavis Elizabeth LLC
- 0228-2820 Hctz 12.5 mg Oral Tablet by Actavis Elizabeth LLC
- 0378-0810 Hctz 12.5 mg Oral Capsule by Mylan Pharmaceuticals Inc.
- 0440-1595 Hctz 25 mg Oral Tablet by Liberty Pharmaceuticals, Inc.
- 0440-7595 Hydrochlorothiazide 25 mg Oral Tablet by Liberty Pharmaceuticals, Inc.
- 0527-1413 Hctz 25 mg Oral Tablet by Lannett Company, Inc.
- 0527-1414 Hctz 50 mg Oral Tablet by Lannett Company, Inc.
- 0527-1635 Hctz 12.5 mg Oral Capsule by Lannett Company, Inc.
- More related products ...
NDC QR Code
Scan the QR code below to easily reference this data in the future:
< Prev: 42708-026Next: 42708-028 >
Related Discussions:
Losartan and Hydrochlorothiazide HCTZ dosing
I am taking hydrochlorothiazide 25mg once a day along with Losartan, also 25 mg. Should I take these together at the sam... 1 reply
I am taking hydrochlorothiazide 25mg once a day along with Losartan, also 25 mg. Should I take these together at the sam... 1 reply
Hydralazine + Hydrochlorothiazide, HCTZ Uses and side effects
My doctor gave me this drug and I don't understand what it is for? What the side effects? Please describe them and t... 4 replies
My doctor gave me this drug and I don't understand what it is for? What the side effects? Please describe them and t... 4 replies
Weight loss from Hydrochlorothiazide, HCTZ + Triamterene
What is the most weight lost by anyone on this drug? I have been taking it for about 3 mos. and have lost weight, but I ... 7 replies
What is the most weight lost by anyone on this drug? I have been taking it for about 3 mos. and have lost weight, but I ... 7 replies
Hydrochlorothiazide 12 5 Mg Side Effects
how do you take the med???and how late to take not to make you get up during the night???? ## When I was on them, my doc... 9 replies
how do you take the med???and how late to take not to make you get up during the night???? ## When I was on them, my doc... 9 replies
Hydrochlorothiazide/25mg effects?
What are the effects of this drug? Are there any side effects? ## half the 25 mg prescribed for HBP; need to know about ... 8 replies
What are the effects of this drug? Are there any side effects? ## half the 25 mg prescribed for HBP; need to know about ... 8 replies
Hydrochlorothiazide 25 Mg Side Effects
After after taking hydrochlorothiazide 25mg I HAVE RINGING In ears. Dry mouth, hearing loss. Not sure if this is related... 6 replies
After after taking hydrochlorothiazide 25mg I HAVE RINGING In ears. Dry mouth, hearing loss. Not sure if this is related... 6 replies
Hydrochlorothiazide And Triamterene By Sandoz Pharmaceuticals
My aunt has been taking the Sandoz Pharmaceutical, hydrochlorothiazide and triamterene for 13 yrs. The GG606, now the ph... 6 replies
My aunt has been taking the Sandoz Pharmaceutical, hydrochlorothiazide and triamterene for 13 yrs. The GG606, now the ph... 6 replies
HYDROCHLOROTHIAZIDE Missed Dose
If you do not take this medication for 3-5 days can it cause water retention right away? ## Yes,I agree,especially durin... 6 replies
If you do not take this medication for 3-5 days can it cause water retention right away? ## Yes,I agree,especially durin... 6 replies
Hydrochlorothiazide 25mg Uses
Is this a water pill or a high blood pressure pill? ## It actually lowers blood pressure by expelling water from the bod... 4 replies
Is this a water pill or a high blood pressure pill? ## It actually lowers blood pressure by expelling water from the bod... 4 replies
Hydrochlorothiazide 12.5mg Capsule
Need to Verify identification of Capsule: Small White Capsule with imprint Mylan 810 ## Pill Image You are correct, this... 4 replies
Need to Verify identification of Capsule: Small White Capsule with imprint Mylan 810 ## Pill Image You are correct, this... 4 replies
Note: The RxChat NDC Database uses publicly available data from the FDA and the U.S. National Library of Medicine (NLM); The NLM is not responsible for the data presented and does not endorse or recommend this or any other product. While we make every effort to ensure that the information presented is accurate, you should assume that all results are unvalidated. To report any errors or inconsistencies please contact us.