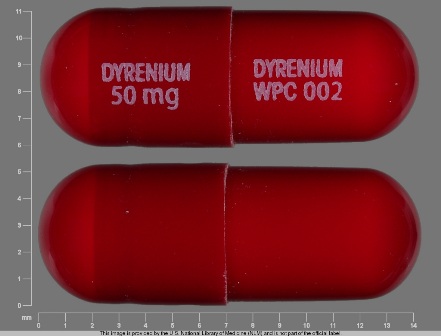
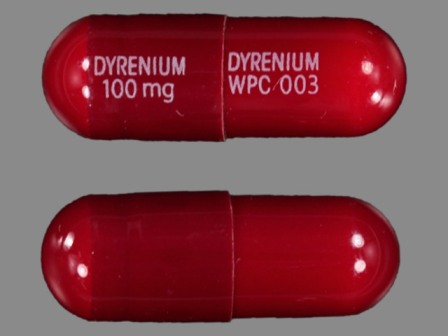
Dyrenium
Active Ingredient(s): TriamtereneFDA Approved: * August 10, 1964
Pharm Company: * WELLSPRING PHARM
Category: Diuretic
Triamterene (trade name Dyrenium among others) is a potassium-sparing diuretic often used in combination with thiazide diuretics for the treatment of high blood pressure or swelling. The combination with hydrochlorothiazide, is known as hydrochlorothiazide/triamterene. Contents 1 Side effects 2 Caution with certain disease states 3 Mechanism of action 4 With hydrochlorothiazide 5 History 6 Research 7 References 8 External links Side effects Common side effects may include a depletion o... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.Dosage List