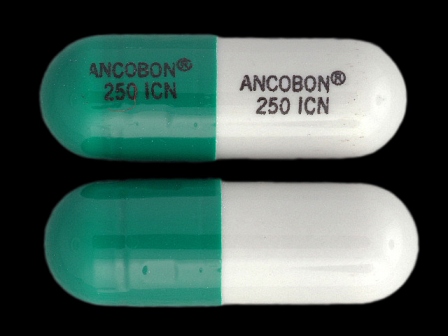
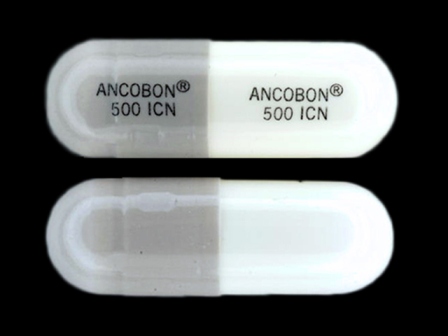
Ancobon
Active Ingredient(s): FlucytosineFDA Approved: * November 26, 1971
Pharm Company: * VALEANT
Category: Antifungal
Flucytosine, also known as 5-fluorocytosine (5-FC), is an antifungal medication.[1] It is specifically used, together with amphotericin B, for serious Candida infections and cryptococcosis.[1] It may be used by itself or with other antifungals for chromomycosis.[1] Flucytosine is used by mouth and by injection into a vein.[1][2] Common side effects include bone marrow suppression, loss of appetite, diarrhea, vomit... [wikipedia]
* May have multiple approval dates, manufacturers, or labelers.Dosage List